CryoCase: Rethink the standard. Replace the bag.

Discover the groundbreaking CellSeal® CryoCase™, the first rigid primary container engineered for closed-system fluid processing in cell and gene therapy (CGT) applications. Launched in November 2024, the CryoCase offers unmatched fracture resistance, transparency for quality inspections, and automation-ready design, addressing the common pitfalls of traditional cryobags and cryovials. With proven performance in freeze-thaw processes and enhanced contamination control, CryoCase is revolutionizing CGT manufacturing workflows. Explore white papers, webinars, and case studies to see how CryoCase can transform your storage solutions. Learn more or request your test sample today!
How Does a Water-Free Automated Thawing System Improve Your Cryopreservation Process?

Cryopreservation has come a long way… Over the past 50 years, advances in biopreservation techniques have enabled industry-wide progress in a range of fields including cell biology research, drug discovery, biobanking, assisted reproduction, and even plant and animal conservation, with the fields of cellular therapy and regenerative medicine seeing the greatest expansion recently. As these […]
Overcoming Cryopreservation Challenges

Navigating the Challenges of Cryopreservation in Cell-Based Research Biopreservation, and specifically cryopreservation, is critical for maintaining the viability of cells long-term throughout storage and research. Recently, BioLife Solutions partnered with Cell and Gene Therapy Insights to produce a poster that highlights many of the challenges researchers face, balanced with Biopreservation Best Practice suggestions. To receive […]
The History of Cryopreservation

Cryopreservation: From Home-Brew to Cutting-Edge Solutions In the pursuit of medical breakthroughs, technology often emerges from the drive to fulfill critical needs. This is particularly evident in the field of cryopreservation, where a convergence of technological advances seeks to preserve therapeutic cells for a myriad of applications, from treating human afflictions to aiding animals in […]
Universal Cell Management System – What is it? and Why is it important to the Cell and Gene Therapy (CGT) Industry?

The Signata CT-5 is a Universal Cell Management System designed for cell and gene therapy production. It enhances efficiency and flexibility in key processes like final fill, media formulation, bioreactor fill, and cell culture wash. With its closed-automation and compatibility with a wide range of consumables, the CT-5 supports scalable, contamination-free cell therapy manufacturing.
Empowering Your Downstream Cell and Gene Therapy Processes: A Sexton Biotechnologies Focus

BioLife Solutions acquired Sexton Biotechnologies to better serve the needs of cell and gene therapy (CGT) developers and manufacturers. Sexton complements BioLife’s existing portfolio by offering media to improve cell performance and cell expansion, automated and flexible fluid transfer manufacturing equipment, and modern CGT storage containers for closed-system processing. Sexton tools can advance your downstream […]
How to Choose the Right Freeze Media
The process of cryopreservation, which has been carried out for decades, is a crucial aspect of cell preservation for various scientific endeavors — from experimental research to clinical applications. While the fundamental process involves freezing cells and storing them at ultra-low or cryogenic temperatures, the choice of cryopreservation media plays a pivotal role in ensuring […]
The Evolution of Cell and Gene Therapy Processing Tools

01/ Borrowed tools from other industries Prior to the invention of gene therapy research, the removal, storage and reinsertion of human cell tissue was limited to blood. The logistical and technical challenges were minimal. However, scientists quickly discovered that maintaining the integrity and transparency of autologous and allogeneic cells throughout extraction, storage, transportation, delivery and […]
Sexton Biotechnologies to be Acquired by Biolife Solutions
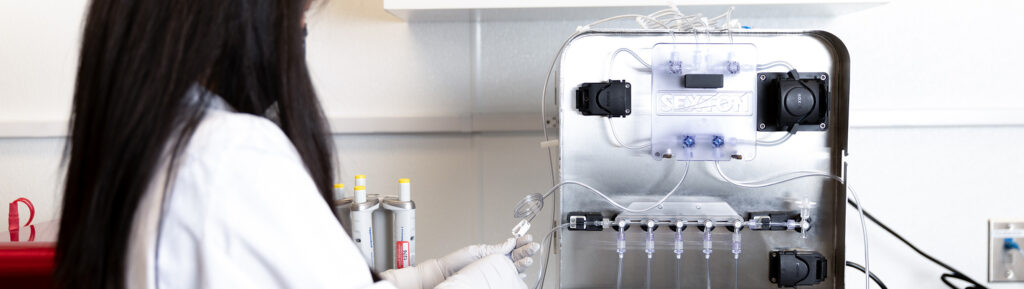
Sexton Biotechnologies, a provider of novel manufacturing solutions for the cell and gene therapy (CGT) industry, today announced that it has entered into a definitive agreement whereby BioLife Solutions (NASDAQ: BLFS) will acquire all outstanding shares of the company in a deal worth a total value of $30 million. During the past two years, Sexton has worked to provide […]
Three Cold Chain Obstacles to Rapid COVID-19 Biologics Distribution
